Understanding Total Dissolved Solids (TDS)
_English_v1_00139.jpg)
Total Dissolved Solids (TDS) is a metric used in water treatment that measures all dissolved minerals, salts, chlorides, metals, organics, and many other contaminants in water. TDS impacts water conductivity and is related to corrosion, chemical efficiency, water clarity, and is an often-forgotten factor in the LSI.
Covered in this article:
- Recommended TDS levels
- What substances contribute to TDS?
- How to measure TDS
- How does TDS impact water chemistry?
- TDS and LSI
- TDS and corrosion
- How to reduce TDS
- Conclusion
Recommended TDS levels
Freshwater Pools
Depending on the source you read, TDS levels should be no more than about 1500 ppm above the tap water's TDS.1 Of course, this requires testing the tap water to know. TDS levels are ideally as low as possible after you have adequate calcium carbonate saturation, which we determine using the Langelier Saturation Index (LSI).
Since TDS includes calcium hardness and alkalinity, keep in mind that TDS below something like 500 ppm is unlikely. Most sources agree that the ideal TDS would be somewhere between 500 and 2500 PPM in freshwater pools.2
Related: LSI Balance and Calcium Management (Pillar 1)
Saltwater Pools
Saltwater pools have a different baseline. Since salt chlorine generators require somewhere in the neighborhood of 3000 to 3500 ppm of salt, the freshwater recommended limit of TDS goes out the window. In our opinion, it brings into question the reason for the limit on TDS in the first place. Our understanding is that TDS serves as a proxy for other factors that may be detrimental to water quality over time. In other words, it gives us a general idea of the "age of the water". And in that sense, TDS is a valuable metric to know.
Perhaps the real question should be "at what level does TDS become a problem?" It is a valid question that is certainly up for debate. The case has been made TDS itself is not really a problem, since the vast majority of it is usually just salts (like sodium chloride) and minerals like calcium. Chlorine is still effective at high TDS levels:
"Total dissolved solids (TDS) naturally elevates as the CYA and the sodium chloride build. As the stabilizer accumulation becomes excessive, TDS is often blamed for a noticeable and sometimes severe drop-off in oxidation and disinfection. Unacceptable cloudiness, algae and even bad health-department reports can be the result, but CYA is held blameless as fingers continue to point at TDS.
“Oh my gosh, you’re nearing 3000 ppm TDS; drain your pool!” is a common cry. Don’t buy this one. As reported in PrP #1 (What’s All This Fuss Over TDS?), high TDS has been inappropriately maligned for as long as CYA’s been around. 10,000 ppm and much more can be harmless in public pool water, as the predominant constituent of TDS in all “aged” pool water is common, everyday salt. In non-stabilized pools, lofty TDS allows perfectly satisfactory chlorine effectiveness. Even chlorine in sea-water pools, at 33,000 ppm TDS, works just fine! Published statements that even 50% reduction in chlorine’s effectiveness occurs when TDS rises to “2000 ppm or 1000 ppm above the make-up water” are patently false." - Kent Williams3
Still, textbooks like the IPSSA Intermediate Training Manual stick with the 1500 ppm above the baseline.4 In the case of a saltwater pool, the baseline is ([tap water TDS] + [3000 to 3500 ppm of salt]). Add 1500 to that baseline, and your new maximum can be as high as 5000 or even 6000 ppm.
In drinking water, elevated levels of TDS can make the water taste worse, according to a World Health Organization (WHO) study.5 But taste should not be a primary driver in swimming pool chemistry. In our opinion at Orenda, we have found no evidence to suggest that TDS levels up to 6000 ppm are detrimental to water clarity or chlorine efficacy. Some sources say TDS can exceed 10,000 ppm, but who really knows? What we do know is that high TDS impacts water balance, and we will discuss that later in this article. What really matters is what dissolved solids make up your TDS level.
What substances contribute to TDS?
TDS is dominated by salts and minerals (primarily sodium and calcium), but also includes chlorides, metals, ions like bicarbonate and carbonate alkalinity, sulfates, nitrogen compounds, and organics. Pretty much anything dissolved in water. Of that list, dissolved heavy metals are usually the smallest contributor to TDS.6
"Total dissolved solids (TDS) is just what it says, the sum of all solids dissolved in the water. The constituents that make up TDS are varied and, for the most part, remain unknown as we simply measure the gross sum of these metals, minerals and salts. Calcium and sodium compounds dominate, while magnesium and dozens of other solubles can be founds therein." - Kent Williams7
We have another article about evaporation and accumulation, which directly applies to TDS. Outdoor pools in climates like southern California and Arizona can evaporate approximately 8-10 feet of water each year. That's more than the average backyard pool, so right there, evaporation at least doubles the TDS in a year.1 And that assumes you did not add any chemicals! But of course, we all have to add at least some chemicals to our pools, even if you are following the Orenda Program of minimalism,
Pool chemicals and their byproducts
Just about every pool chemical you can think of leaves behind something that contributes to TDS. Let's do a brief rundown of the common pool chemicals.
- All types of chlorine, when reduced (used up), become chloride (Cl-), which is basically a salt.
- Liquid chlorine leaves behind sodium and chloride, which is common salt (NaCl).
- Cal Hypo leaves behind about 4 ppm / pound / 10,000 gallons of calcium, as well as additional chloride.
- Dichlor and Trichlor both leave behind cyanuric acid and chloride.
- pH adjusters like sodium bicarbonate and sodium carbonate leave behind sodium (obviously) and alkalinity ions.
- Algaecides, depending on their type, leave behind byproducts like nitrogen compounds, copper, etc.
- Calcium chloride (CaCl2) directly adds to TDS with both calcium and chloride.
Hopefully, you get the idea. Almost everything adds to TDS. However, there are also some chemicals that are capable of reducing TDS slightly–like CV-600 enzymes that remove organics–but the TDS reduction is usually negligible. We will cover how to lower TDS later in this article.
How to measure TDS
TDS is measured using an electronic TDS meter or a test strip. Most electronic TDS meters measure the conductivity of water (not to be confused with ORP, which measures the oxidation-reduction potential in mV). Electrical conductivity increases with higher TDS, and a digital probe can easily tell what that conductivity is. Proper maintenance and calibration of any TDS instrument is critical to the accuracy of the test. With a properly calibrated instrument, test results can be quick and precise.
Additionally, test strips are available for a quick measure of TDS. A simple, quick check of the water using strips can provide guidance on the TDS levels. Users can quickly determine if TDS is above the recommended range, though results may not be as precise as the electronic options.
How does TDS impact water chemistry?
 Some sources online claim TDS impacts chlorine efficiency and leads to cloudy water, scale formation, and all sorts of other problems. Perhaps all of that can be true, but it depends on the circumstance. Take cloudy water, for example. Total Dissolved Solids means the solids are dissolved in water, not suspended and floating around. Truly dissolved solids are invisible because they are in solution. In other words, TDS alone does not create cloudy water. But TDS can be a factor that leads to cloudy water.
Some sources online claim TDS impacts chlorine efficiency and leads to cloudy water, scale formation, and all sorts of other problems. Perhaps all of that can be true, but it depends on the circumstance. Take cloudy water, for example. Total Dissolved Solids means the solids are dissolved in water, not suspended and floating around. Truly dissolved solids are invisible because they are in solution. In other words, TDS alone does not create cloudy water. But TDS can be a factor that leads to cloudy water.
Look at the photo above of the underwater light. All those particles floating around are not dissolved...they are suspended. Therefore, we can see them, and they cannot be counted toward the TDS number. Cloudy water is usually caused by filtration issues or non-living organics and the debris that it can bind to. That, or an LSI violation, which causes calcium carbonate to precipitate out of solution.
The real impact of TDS is found when using the LSI.
TDS and the LSI
The Langelier Saturation Index (LSI) is the objective way to measure water balance. It is a calculated index that tells us how saturated water is calcium carbonate (CaCO3). There are six factors that go into the calculation for swimming pools. They are:
- pH
- Carbonate Alkalinity
- Calcium Hardness
- Water Temperature
- Cyanuric Acid (CYA)
- Total Dissolved Solids (TDS)
The higher the TDS, the lower the LSI. We know that sounds backwards, but it is not. TDS has an inverse relationship to the LSI. Higher TDS means more aggressive water. While most freshwater pools have TDS levels below 2000, their impact on the LSI is pretty small. But the higher you go, the faster the LSI drops. See the screenshot from the Orenda app:
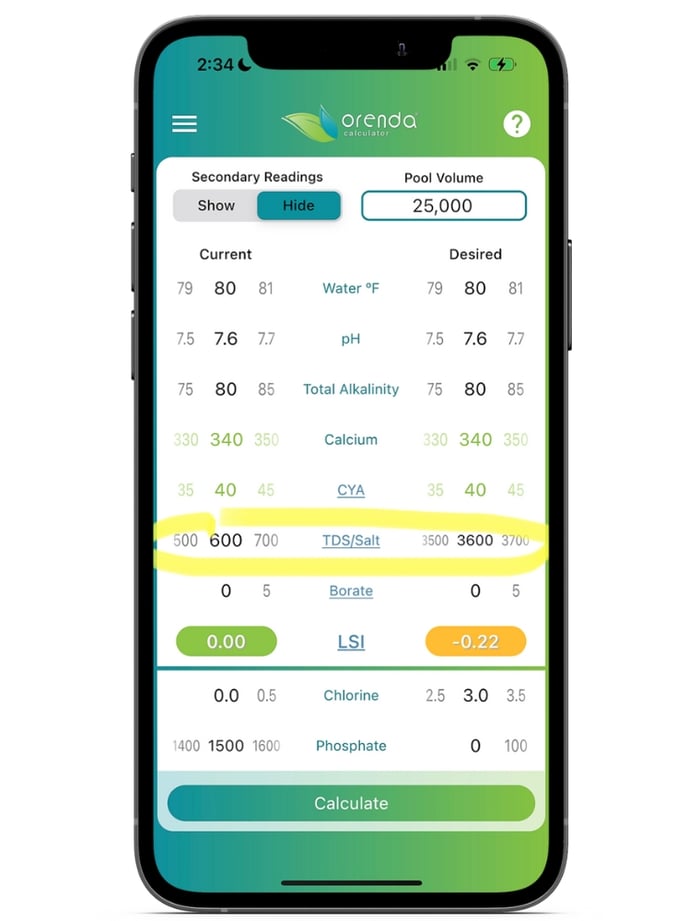 By simply making this balanced pool (on the left) into a saltwater pool (on the right), the water went from balanced to almost aggressive. And by the way, this screenshot was just an illustration of what salt alone does. For actual TDS, we need to include our alkalinity and calcium hardness in addition to salt. Keep that in mind when using the Orenda App. The LSI formula calls for TDS, which includes salt and everything else.
By simply making this balanced pool (on the left) into a saltwater pool (on the right), the water went from balanced to almost aggressive. And by the way, this screenshot was just an illustration of what salt alone does. For actual TDS, we need to include our alkalinity and calcium hardness in addition to salt. Keep that in mind when using the Orenda App. The LSI formula calls for TDS, which includes salt and everything else.
TDS and corrosion
While the LSI talks about etching and scale formation, corrosion itself is not always an LSI consequence. Remember, TDS is generally measured with a conductivity meter, meaning there is more potential for electrical conductivity. Therefore, high TDS levels increase the risk of metal corrosion in a swimming pool.8 This, as you probably already know, is magnified in a salt pool, especially if it was not bonded 100% correctly during construction. Any difference in charge can lead to accelerated degradation of stainless steel or other metal components like screws.
How to reduce TDS in a swimming pool
Reducing TDS is done the same way you reduce salt, calcium hardness, nitrates, or cyanuric acid. Drain and dilute. If draining is not an option for you–like water restrictions during a drought–then reverse osmosis filtration is a great option.
But TDS alone is almost never a primary reason to drain. Sure, several textbooks and online forums say to drain when you have 1500 ppm over your baseline, but we have not found enough evidence to support that. Our field experience indicates higher TDS is not problematic until the LSI becomes compromised. Draining and diluting becomes necessary when LSI balance is too difficult to maintain and/or the pool is overstabilized with too much cyanuric acid.
Related: How to Reduce CYA in a Swimming Pool
Conclusion
Total Dissolved Solids are primarily made up of salts and minerals. It also includes chlorides, sulfates, ions of alkalinity, nitrogen compounds, and non-living organics that are dissolved in water. TDS does not directly contribute to cloudy pool water, but it can lead to circumstances where water becomes cloudy. TDS is usually measured using an electronic conductivity meter but can also be measured using a test strip. It is an often-overlooked factor of the LSI. Higher TDS means lower LSI.
TDS itself is not necessarily a problem until maintaining LSI balance becomes too difficult. If it gets to that point, drain and dilute to lower your TDS level.
1, 2 Lowry, Robert. (2016). IPSSA Basic Training Manual, Part 1 - Chemicals. Pages 43-44.
3 Williams, Kent. (1997). Cyanurics: Benefactor or Bomb?
4 Lowry, Robert. (2009). IPSSA Intermediate Training Manual, Part 1 - Chemicals. Page 114.
5 Originally published in Guidelines for Drinking Water Quality. (1996). Health criteria and other supporting information. World Health Organization. (2nd ed. Vol. 2)
6 NSF International Consumer Fact Sheet. Total Dissolved Solids in Drinking Water.
7 Williams, Kent. What's the Fuss over Total Dissolved Solids? PPOA Pumproom Press.
8 APSP Recreational Water Quality (RWQ) Committee. (2015). Total Dissolved Solids. APSP Fact Sheet.
