Understanding Vinyl Liner Pools

Swimming pool vinyl liners are a popular alternative to cement-based finishes and fiberglass pools. At Orenda, the majority of our focus has been on cement-based finishes (plaster, quartz, pebble, etc.) because of their interaction with water chemistry–especially during the initial few days of a startup. But this article is specific to vinyl liners and how they interact with the water. Spoiler alert: the LSI still matters.
Covered in this article:
- Vinyl liner overview
- Liner standards
- Vinyl liner strengths and weaknesses
- Strengths
- Weaknesses
- The LSI and vinyl liners
- Fading and deterioration
- Absorption and wrinkling
- How to treat a vinyl liner pool
- In season chemistry
- Out of season (and winterization)
- Conclusion
Before starting, this article is not an opinion piece. It is meant to inform by showcasing pros and cons, but focuses on water chemistry considerations.
Vinyl liner overview
Vinyl liners are a popular type of pool surface. They are custom-made to fit your specific pool and a trained installer puts them in place. Because they are pre-fabricated and installed relatively quickly, vinyl liner pools are usually less expensive to build and renovate than in-ground gunite pools with cement finishes. A typical vinyl liner replacement may take only a few hours with an experienced crew (not including water fill time), as opposed to days or weeks for a renovation and re-plaster. If you have never seen a vinyl liner installation, it's pretty neat to watch. Here's a cool video showing the basics. It should go without saying that this work should be done by trained professionals:
Liner standards
Swimming pool liner standards are established by the Chemical Fabrics and Film Association (CFFA).1 The most common thicknesses for residential liners are either 20 or 28 mil. Commercial pools use much thicker 60 mil (1.5 mm) liners.2 60 mil liners are less common in residential pools. And if you don't know what "mil" means, you are not alone. We had to look it up. One 'mil' is 1/1000th of an inch.3
Vinyl liner strengths and weaknesses
There has been extensive research done by some of the original vinyl manufacturers, such as Canadian General Tower (CGT), and we want to summarize some of it here for you. We found much of it valuable and worth sharing.
Strengths
Liners are widely available, more affordable to install and replace, and can be installed quickly. They have benefits especially in colder climates, due to the lack of winter issues like calcite crystals. Even if a vinyl liner is damaged and faded during the winter, its surface is still smooth and therefore not abrasive to swimmers' feet. Another benefit is that patterns and colors do not drive the price of a liner. You can choose from many patterns and colors without large differences in cost, unlike cementitious surfaces which can vary widely in price.
Liners also do not interact as rapidly with water chemistry as a cement-based finish (plaster, pebble, etc.) does. Therefore liners have less urgency for chemical balance on a startup, and less likelihood of a spiking pH. In other words, vinyl liner pools should be easier to maintain water balance in. If you're following the LSI, liners offer more grace and time to get your chemicals in order. All in all, vinyl liner pools tend to be easier to manage and more affordable.
Expanding on that same train of thought, if Trichlor is your primary chlorine, pH tends to stay too low in vinyl liner pools compared to plaster pools. This is because Trichlor suppresses pH (because it is acidic chlorine), and the vinyl surface offers nothing to artificially raise pH again. This is good–in the sense that you're less likely to damage a vinyl liner pool in the short term. But it can also be a weakness if you are used to pH rising. It is important to maintain LSI balance and not go too low on your pH and alkalinity in a vinyl pool if using Trichlor.
Weaknesses
Many would argue the biggest weakness of vinyl liner pools is their lack of customization. Concrete pools can be just about any shape and size, whereas liners are more limited in their shape and sizes. That all being said, technology continues to advance and we have been seeing more complex liner pools out there. In that same vein, while you can get a plethora of patterns and colors, vinyl liners are almost all a similar feel and appearance...unlike a concrete pool which can have many textures and vibrant colors.
Believe it or not, vinyl liners can actually contain trace amounts of calcium in them.4 This surprised us too! And it brings into question why most recommendations for vinyl pool chemistry have calcium lower than in cement surface pools. Most vinyl manufacturers recommend between 150-250 ppm calcium hardness. But now that we know calcium carbonate is in the material itself, problems like fading, porosity, and wrinkling start making a lot more sense. The LSI still matters in vinyl pools! In our opinion, range chemistry recommendations–regardless of pool type–should always be followed within the context of the LSI first.
The weakness is not that vinyl liners contain calcium carbonate (CaCO3). The weakness is that they contain so little of it that the water hardly has any means of correcting itself if the LSI gets too low (below -0.30). Sure, this delayed urgency could also be viewed as an advantage, but we view it as a disadvantage. This means that aggressive water stays aggressive longer. Think about a winterized liner pool. Cold, stagnant water...getting colder and colder as the weeks go on in the fall. How is the water going to saturate itself with the calcium it needs if you don't winterize the pool properly? Well, according to CGT's research, water can absorb into the vinyl (more on this in a moment). We presume water penetrates in pursuit of that precious calcium carbonate. This leads us to our next subject.
Related Procedure: How to Winterize a Pool, The Orenda Way
The LSI and vinyl liners
As mentioned before, water balance absolutely matters for pools with vinyl liners. Sure, you may have more time to act before you see problems like fading, deterioration or wrinkling. But rest assured, if the LSI is too low, the damage is occurring.
We disagree with the recommendations for 150-250 ppm of calcium hardness because...
- that recommendation does not factor in the LSI (especially water temperature),
- the alkalinity and pH recommendations allow for only a narrow window of water balance within that given calcium hardness range, and
- because in most parts of the United States, 150 ppm of calcium is just too low, period. You can find out for yourself while playing with the Orenda Calculator™. Good luck maintaining LSI balance with just 150 ppm calcium hardness when the water drops below 75ºF.
Here are some of the issues that can happen if you ignore the LSI:
Fading and deterioration
Most people we talk to think that fading and liner deterioration is the result of over-chlorination, or a low pH, or our favorite, a blanket statement like "bad water chemistry". It's not that those answers are wrong, it's that they are too generalized and not specific enough. pH being too low is a problem because it lowers the LSI. Perhaps there is some truth to the over-chlorination idea, as illustrated in CGT's research,5 but is it as significant as aggressive water gnawing away at the liner, and pigment being collateral damage? Our research [so far] tells us no. The LSI is a far more important issue.
Absorption and wrinkling
Of all the things we have learned since starting to research vinyl pool liners, two things, in particular, surprised us. First, as mentioned before, vinyl liners often contain calcium carbonate. Second, vinyl liners can absorb water. And here we always assumed they were 100% waterproof. Liner porosity leads to bigger problems like wrinkling and fading.
The bottom line: The LSI matters, and because there is so little CaCO3 for the water to take, the consequences can be more severe over time. Water can stay aggressive for longer, and permanent damage to the liner can occur.
How to treat a vinyl liner pool
We are often asked how to manage water chemistry in vinyl liner pools, and if liner pool chemistry needs to differ from in-ground gunite pools. The answer is yes, liner pools (and fiberglass pools) have some different needs than pools with cementitious surfaces. That being said, the goals for water balance are the same, and sanitization itself is identical.
Just like pools with cement finishes, water temperature throughout the year will drive much of your LSI chemistry strategy. Temperature cannot be ignored. So let's divide our strategy in two: in season, and out of season.
In season
If your primary chlorine has a high pH (liquid sodium hypochlorite or calcium hypochlorite), you will want your alkalinity around 70-80 ppm in the summertime. This will help limit how high your pH can rise naturally. To offset this lower alkalinity, you will need more calcium hardness. Use the Orenda App to figure out where your calcium and alkalinity levels should be, factoring in all six components of the LSI.
If you have a salt system, your alkalinity should be even lower, between 60-70 ppm. Here again, you need to have higher calcium hardness to offset this! When you have lower alkalinity, it takes less acid to reduce pH, so use the dosing calculator to factor that in! Do not eyeball chemicals and assume you know how much acid it takes to reduce the pH. Learn more about correcting these bad habits here.
If your primary chlorine is acidic, like Trichlor, you may want 90-120 ppm alkalinity, because pH will be suppressed by your chlorine.
Out of season (and winterization)
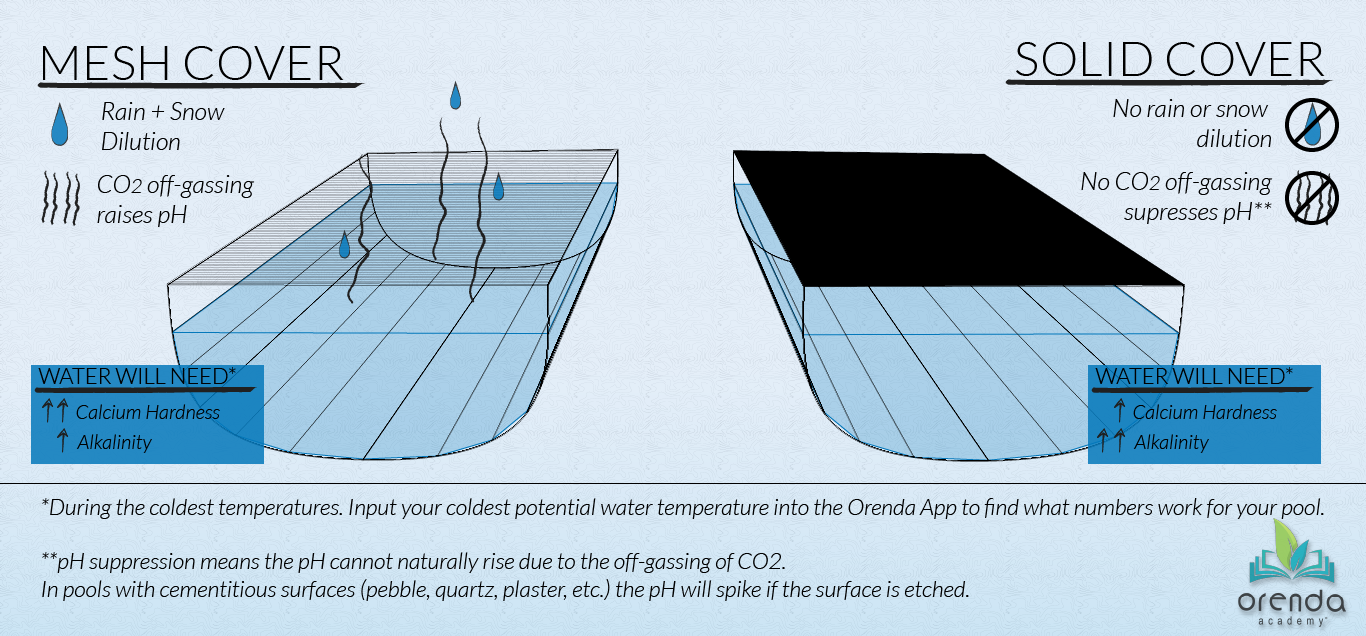
When water gets cold, the need for calcium rises. Winterize with the future coldest temperature in mind. If you know your pool will freeze, prepare for it and let the pH naturally rise up to its ceiling, (usually about 8.1-8.2). You may need to increase alkalinity to get through the winter and make sure that at the coldest temperature, your LSI remains balanced in those conditions.
The type of safety cover you have also matters to winterization chemistry. See the diagram below.
Conclusion
This became a longer article than we intended. Sorry. You know how it goes...start with one idea and the research rabbit hole opens up. To summarize:
- Vinyl liner pools often require less maintenance of water chemistry due to less interaction between water and surface. But the chemistry still matters!
- Vinyl liners contain small amounts of calcium carbonate (CaCO3), making the LSI even more important than we originally thought. We always knew aggressive water could fade liners and increase porosity, and this explains why.
- Pool liners have advantages and disadvantages, but if you maintain your water correctly, you can prevent most of the problems from occurring, and extend the life of the liner.
- Especially in cold water, liners can face prolonged damage because the water has almost no mechanism for correcting its chemistry if it goes aggressive. Especially if you have a solid safety cover over it that prevents CO2 from off-gassing and raising the pH.
Vinyl liners are great pool surfaces, but it is up to you, the pool owner or service technician, to treat the water right.
1 CFFA Standards for vinyl liners, 2017
3 "Mil" is not short for millimeters (28 millimeters would be about an inch thick!). Mil is one one-thousandths (1/1000th) of an inch thick. So 28 mil is 28/1000ths of an inch thick. So a 28 mil liner is less than one millimeter thick (0.7112 mm).
4 We first heard about vinyl-containing calcium carbonate from someone who asked an online search engine about it. That led to Wikipedia and other sources talking about vinyl for other uses–particularly flooring. And yes, it turns out that calcium carbonate is regularly used as a filler to supplement Poly (vinyl chloride). Who knew?
5 We tried to hyperlink to the actual technical bulletin we are citing, but their website has all of them in succession with anchor links. So to find what we're talking about, go here, then tap the "chlorine levels" button.
