The Four Pillars of Proactive Pool Care

Are you proactive in your approach to pool management and water chemistry, or are you reactive? Based on the types (and amounts) of products sold in the pool business, it is clear that plenty of pool people are reactive. According to Dr. Charles Bens, less than 10% of healthcare costs go toward proactive measures...which means over 90% is spent reacting to diseases and illness. In other words, over 90% of the medical industry is treating symptoms and consequences, not preventing them. Amazingly, the same argument could be made in the swimming pool industry.
It is always more cost effective to prevent a problem than to fix it after it starts. The old adage goes something like "an ounce of prevention outweighs a pound of cure", right?
We at Orenda have always had a minimalist, proactive philosophy about water chemistry. Recently, however, we have refined our message and we feel pretty good about how to articulate it now. We call it the four pillars of proactive pool care.
Related: How to Implement the Orenda Program
The Four Pillars of Proactive Water Chemistry
From a water chemistry perspective, there are four fundamental things we focus on. Even if you conquer only one of these four, water chemistry becomes easier to manage, compared to the old way. The four pillars of Orenda pool care are:
- LSI Balance and Calcium Management
- Carbon / Organic (bather waste) management
- Phosphate Removal
- Minimal Cyanuric Acid (CYA, or stabilizer)
This article is only an introduction to each concept, and we encourage you to read the articles referenced here, as they go more in depth on the topics.
Pillar 1: LSI Balance and Calcium Management
ACTION STEP: Maintain LSI balance year-round.
We need to come together and understand how calcium behaves in water. Let's start by debunking a myth in the pool business: calcium is NOT your enemy! It is actually your best friend. Yes! Calcium does amazing things for water chemistry, and helps pool owners and operators balance their pools according to the LSI. The LSI (Langelier Saturation Index) is a measurement of calcium carbonate (CaCO3) saturation in water. Too much (high LSI over +0.30) can lead to calcium falling out of solution as scale. Too little (low LSI below -0.30) means your water is aggressive and looking for calcium to consume. This is the cause of etching, corroded pool equipment and many other problems.
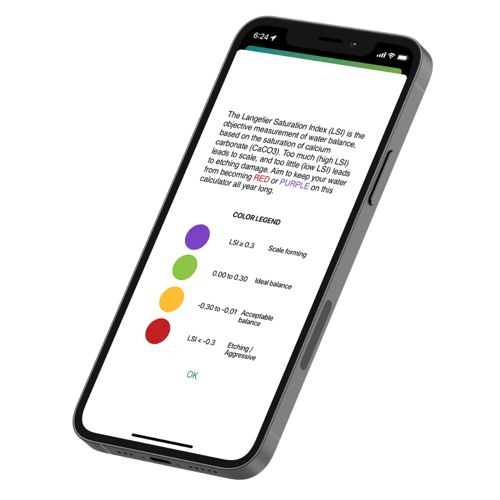
When the pool is properly balanced with calcium, other factors of water chemistry become surprisingly easy to manage. Factors like pH and total alkalinity seem to stay more consistent. Read the articles below about calcium management and the LSI. Understanding how calcium behaves means we can predict and control it. Say goodbye to scale and etching!
- Calcium: The most misunderstood chemistry in the pool business
- Calcium Scale vs. Crystals
- Understanding Calcium Hardness
- Why does Soda Ash sometimes cloud up a pool?
- Three types of Calcium dust in swimming pools
- Four things to do when you open a pool with calcium crystals in it
- Pool winterization water chemistry
- Cold water is more corrosive and can etch plaster
- LSI First, Range Chemistry Second
- How to use the Orenda App: Dosing Calculator + LSI
- Understanding the LSI: Langelier Saturation Index
- How to Prevent Scale in Pool Heaters and Salt Chlorine Generators
- Understanding Total Dissolved Solids (TDS)
Pillar 2: Organic Waste Management
ACTION STEP: Supplement chlorine against the oxidant demand.
Bather waste. Non-Living Organics. Carbon-based crud. Body Butter. People have different names for organic waste in the water, but here's what you need to know: you need to manage it. If you do nothing, chlorine itself will try to oxidize the organics out of the water...and that's great...except chlorine is not a very efficient oxidizer of organics. Chlorine is an excellent sanitizer and disinfectant, yet over 80% of its job in swimming pools is oxidation of bather waste.

Isn't that crazy? That would be like having an excellent salesperson in your company being kept in the office doing paperwork and accounting. In the Orenda program, we know that being proactive against bather waste is essential to water quality and sanitizer efficiency. So we use enzymes to break down non-living organics and devour them. Enzymes can help do the heavy lifting so chlorine is free to do what it does best: sanitize.
More about non-living organics and enzymes:
- How Orenda Enzymes actually work
- Warmup Pool Overloaded: How Orenda Enzymes handled 850 swimmers
- World-Class water clarity: the case for enzymes
- Enzymes. Why treat your water with them?
- Change we can relieve in- A more practical way to address urine in swimming pools
- Refurbishing a sand filter with enzymes vs. doing a sand change
Pillar 3: Phosphate Removal
ACTION STEP: Keep phosphate levels below 500 ppb.
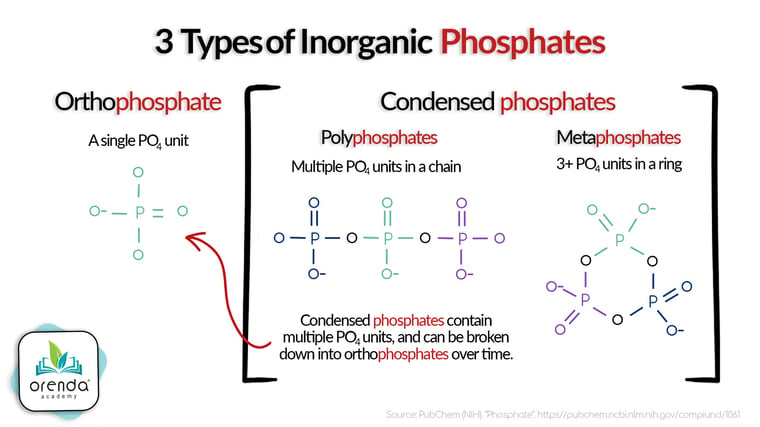
Phosphates are one of several essential micronutrients. Other micronutrients found in swimming pool water include nitrates (and other various nitrogen compounds), and carbon-based nutrients. Phosphates, however, seem to have come out of nowhere in the past decade. Why? Because in most of the country, water treatment practices now include a phosphate-based sequestering agent in drinking water. The sequestering agent helps protect the infrastructure from scale and corrosion, but it means phosphates are in the tap water.
Well water also tends to have high levels of phosphates, and phosphates can be introduced naturally from the weather and other elements around the pool. Removing phosphates can significantly improve water quality and sanitizer efficiency in the process. Learn more about phosphates here:
- Phosphates, Algae and Chlorine Demand
- Green pool, full of algae, but no Phosphates?
- Phosphates in Pool Water
- Wildfires and Swimming Pools
- Different types of Phosphates
- Green Pool Clean Up Procedure
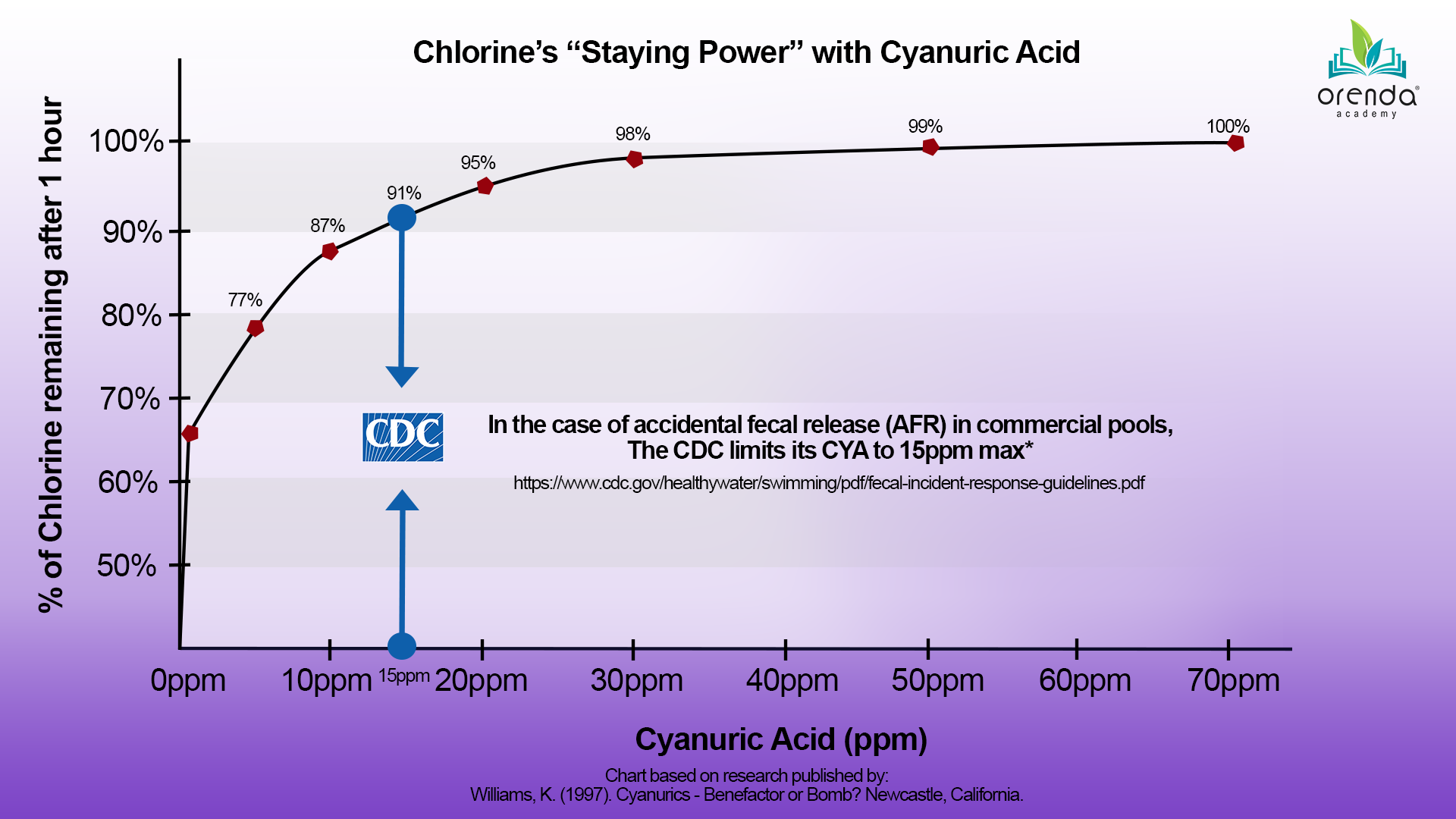
Pillar 4: Minimal Cyanuric Acid (CYA)
ACTION STEP: Do not let CYA levels exceed 50 ppm.
With all we have learned about stabilizer (cyanuric acid, or CYA), it is no surprise that we should keep it to a minimum in our water. Minimal CYA still has tremendous sunlight protection for chlorine. Over-stabilization, as we continue to learn, is an ever-increasing problem. Sanitization and oxidation are affected, as is testing water itself. With high levels of stabilizer (over 50 ppm), maintaining a good chlorine residual becomes nearly impossible. For more about cyanuric acid, read some of our other articles: