Understanding Bromine Pools and Spas

Bromine is the most popular alternative sanitizer to chlorine. They are both halogen elements and neighbors on the Periodic Table. However, the chemistry of bromine differs from that of chlorine, and that's precisely what we'll delve into in this article.
Covered in this article:
- What is a residual pool sanitizer?
- Bromine
- Types of bromine
- Chemistry of bromination
- pH impact on Bromine
- Bromine dosing vs. Chlorine dosing
- Why does Bromine turn water green?
- The Bromine Cycle
- Bromine byproducts
- Bromamines
- Trihalomethanes (THMs)
- Bromates
- What secondary disinfection systems can be used on a bromine pool?
- Is bromine compatible with chlorine?
- Bromine can take over a chlorine pool
- Conclusion
What is a residual pool sanitizer?

A residual sanitizer is a substance that kills microorganisms in water and circulates throughout the swimming pool. A residual sanitizer circulates throughout the pool and its entire circulation system, providing the first line of defense against what the CDC calls Recreational Water Illnesses (RWIs).
A residual sanitizer can be measured on a test kit in parts-per-million (ppm) or its metric equivalent, milligrams-per-liter (mg/L). A secondary sanitizer would be a system like UV, Ozone, or AOP. They are secondary because they do not create a residual in the pool. These systems are point-of-contact.
By far and away, the most common residual sanitizer is chlorine. But this article is about the second-most popular pool sanitizer, bromine. And while they are similar substances, bromine and chlorine chemistry are quite different. And so are their byproducts and health implications.
Bromine

Bromine is more often used in spas and hot tubs than in pools. This is primarily out of convenience because bromine is available in small tablets. These tablets dissolve slowly and work well in warm, aerated water like hot tubs.
Despite bromine being more expensive and weaker than chlorine, it is still popular in spas/hot tubs. Perhaps this is because the bromine byproducts we can smell (bromamines) are less harsh for most people than chlorine byproducts (chloramines) or because bromine tablets dissolve slower in hot water than chlorine tablets. Whatever the reasons, consumer demand keeps bromine tablets on the market for spas.
If bromine is going to be used in a pool, it should only be in indoor pools because bromine cannot be stabilized against ultraviolet (UV) degradation. Unlike chlorine, there is no cyanuric acid equivalent for bromine to use.
Both bromine and chlorine are part of the halogen group of elements on the Periodic Table. Each element in the halogen column is capable of disinfection, and they are known for creating salts when bound to sodium. Sodium chloride (NaCl) is the salt we're all familiar with, and sodium bromide (NaBr) is bromine's salt, which we will explain later in this article.
On the periodic table, bromine is below chlorine, making it less reactive. This concerns the elements' oxidation states (i.e., chlorine is a stronger oxidizer than bromine). Because of this, chlorine can actually oxidize bromide ions into active bromine indefinitely. This is important to understand, and we'll elaborate in a moment.
Types of bromine used in pools and spas
Bromine is sold as tablets, briquettes, granules, and in liquid form. Each of these products is different and has a different pH impact on the water.
According to the Pool Chemistry Training Institute handbook, There are three bromine products used in recreational water:1
- Two-part bromine (sodium bromide + a separate oxidizer)
- Neutral pH because sodium bromide, like salt, does not affect pH.
- Requires a separate oxidizer to oxidize bromide ions into active bromine
- Sodium bromide is a common pool algaecide, but it's really just a salt (like sodium chloride is table salt). Sodium bromide leaves behind bromide ions (Br-) that get oxidized by the separate oxidizer, which then creates Hypobromous acid (HOBr), the active sanitizer. Using this chemical product on a chlorine pool can really easy build the bromine levels until the pool will irreversibly be a bromine pool.
- BCDMH (bromo-chloro-5,5-dimethylhydantoin) tablets or sticks
- 4.5-4.8 pH (moderately acidic due to the HOCl created)
- Chemical formula: C5H6BrClN2O2
- Contains some chlorine, and both HOBr and HOCl will be created
- Most popular bromine product on the market, specifically for spas.
- Does not require a separate oxidizer to work, though beginning with sodium bromide to build a "bromide bank" in the water is recommended when first using this product
- DBDMH (dibromo-5,5-dimethylhydantoin) tablets or briquettes
- ~6.6 pH (relatively neutral)
- Chemical formula: C5H6Br2N2O2
- Chlorine-free
- Also requires a separate oxidizer to oxidize bromide ions into active bromine
The chemistry of bromination
Just like with chlorine, when any of these bromine types dissolve in water, they all produce Hypobromous acid (HOBr), the active form of bromine. This is bromine's equivalent to Hypochlorous acid (HOCl). So when bromine dissolves in water, it looks like this:
Br2 + H2O → HOBr + HBr
bromine + water → Hypobromous acid + Hydrobromic acid
Then the Hypobromous acid (HOBr) dissociates into hydrogen and Hypobromite ion, just like HOCl dissociates into hydrogen and Hypochlorite ion:
HOBr ⥄ H+ + OBr-
Hypobromous acid dissociates into Hydrogen ion and Hypobromite ion
The reason for the (⥄) instead of equal dissociation is that this equilibrium happens at a higher pH than pools and spas normally operate under. So most of the active bromine in water will be HOBr relative to OBr-.
pH impact on Bromine strength
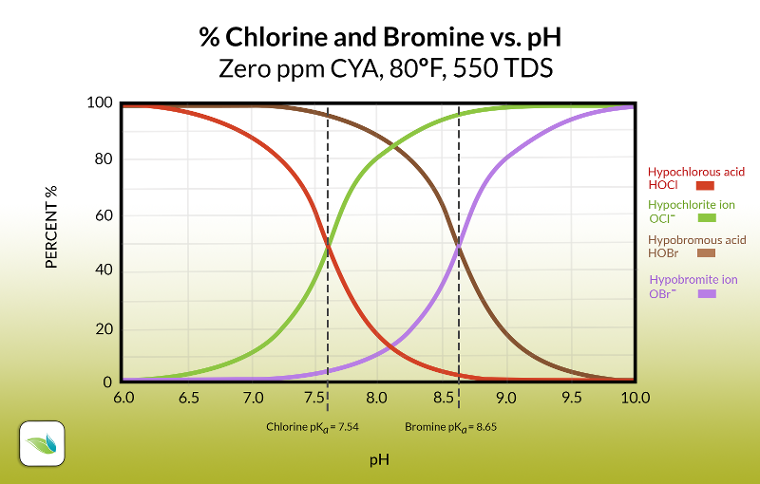
Like a chlorine pool without CYA, the pH controls the percentage of hypobromous acid (%HOBr), representing the "strength" or efficacy of bromine. The lower the pH, the higher the % HOBr; therefore, the faster the contact times (CT) required to kill known pathogens. And because there is no CYA equivalent for bromine, pH always determines the %HOBr in a bromine pool.
pKa values
While chlorine is about 50/50 at 7.54 pH, bromine is 50/50 at 8.65 pH. These specific pH points are called pKa values. We may decide to cover pKa values in more depth in a future article, but for now, know a pKa value is where two species in equilibrium are 50/50 with each other.
This is great news for swimming pools and spas because even at 8.65 pH, you still have 50% HOBr. Any pH below that is an even higher percentage.
Bromine dosing vs. chlorine dosing
Bromine is a weaker disinfectant and oxidizer than chlorine. It is also 2.25 times heavier than chlorine, requiring 2.25 times the chlorine dose (in ppm) to do the same job.2 According to Bob Lowry's textbook, the ideal bromine level is between 4.0 and 6.0 ppm.
Bromine kills germs and algae like chlorine (and other halogens). It attacks cell walls and destroys pathogens from within. It oxidizes the same way too, it's weaker than chlorine, so it takes more for bromine to do the same job.
Why does bromine turn water green?

If you have ever seen a bromine pool or spa, you might have noticed a green tint to the water. The deeper the water, the more noticeable the color, especially when compared to chlorinated water. Brominated water looks green because of the color of the bromine itself. According to Richard Falk:
"Bromine is brown when concentrated, and yellow when diluted. If you have a blue colored pool, then yellow + blue = green. Also, pool water without bromine is bluish if deep enough because the red in sunlight gets absorbed by the water, leaving blue to be reflected and seen. So it's the yellow of bromine plus blue of the water that has the net color look green." - Richard Falk 6
The bromine cycle
And just like HOCl gets reduced to chloride ions (Cl-), HOBr gets reduced to bromide ions (Br-) when it oxidizes or kills contaminants in water. But that's where the similarities to chlorine end.
Without a salt chlorine generator, chloride ions are inert and useless in water. Using electrolysis, salt chlorine generators "recharge" chloride ions into chlorine gas (Cl2). But bromide ions can be oxidized into active bromine as Hypobromous acid (HOBr). That HOBr will continue oxidizing and disinfecting water. Because bromine is beneath chlorine on the periodic table, chlorine can oxidize and "recharge" bromide ions.3 The system in basic terms looks like this:
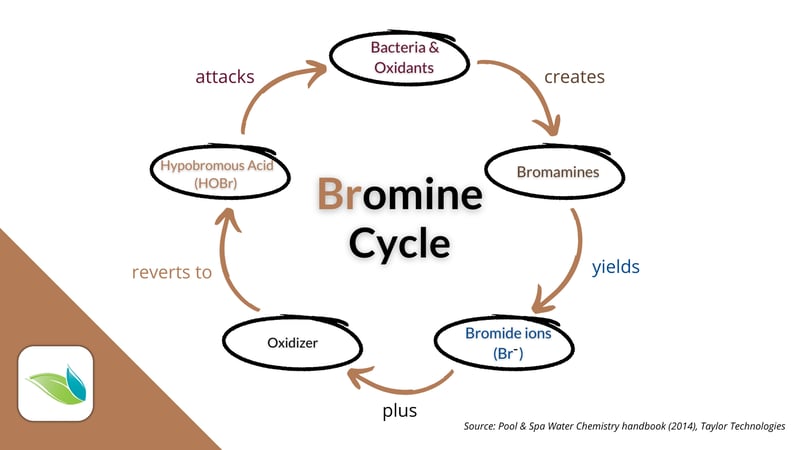
And it's not just chlorine that can oxidize bromide ions back to active bromine. Secondary oxidizers like ozone and AOP can also do it, as well as a non-chlorine oxidizing shock like potassium monopersulfate.4
But as we'll explain later in this article, secondary systems are not the best options for bromine pools for a different reason. To resume the differences between chlorine and bromine we can establish the following facts:
- Chloride ions (Cl-) are inert unless recharged by electrolysis in a salt chlorine generator. Electrolysis creates chlorine gas (Cl2) and other byproducts.
- Bromide ions (Br-) can be recharged by any oxidizer, including chlorine, potassium monopersulfate, Ozone (O3) and hydroxyl radicals (•OH).
So while you don't necessarily need to buy a lot of bromine, keeping a bromine pool active costs more money than a chlorine pool because of the constant need for an oxidizer.
Bromine byproducts
Bromamines
Bromine attacks and destroys germs and oxidants. And like chlorine, bromine is inefficient at oxidizing non-living organics and oils, so it should be supplemented against the oxidant demand, especially in high-bather water, like spas and hot tubs.
But unlike chlorine, total bromine and free bromine are not distinguished ("combined bromine" is not measured because it does not need to be). Just measure total bromine. And that's okay because bromamines are decent sanitizers, so there's really no reason to distinguish them in a test.
Bromamines are created when bromine combines with nitrogen compounds as part of the oxidation process.5 Then, they are destroyed when enough HOBr can overwhelm the bromamines. HOBr and bromamines will yield bromide ions (Br-), and the bromine cycle continues.
Bromamine formation
HOBr + NH3 ⇌ NH2Br + H2O
Hypobromous acid + ammonia ⇌ monobromamine + water
HOBr + NH2Br ⇌ NHBr2 + H2O
Hypobromous acid + monobromamine ⇌ dibromamine + water
HOBr + NHBr2 ⇌ NBr3 + H2O
Hypobromous acid + dibromamine ⇌ tribromamine + water
Bromamines (like chloramines) have some disinfection power and are eventually reduced to bromide ions (Br-). HOBr can also get reduced when it kills germs or algae. These bromide ions–known as the bromide bank–can then be oxidized by a chlorine shock, non-chlorine shock, or secondary oxidation system to create HOBr once again.6 Then, the cycle repeats itself.
Some sources say that bromamine byproducts do not cause irritation or smell bad. But that's a matter of opinion because indoor bromine pools certainly have a distinct smell. Bromine is noticeably less harsh than chlorine but can seem worse to some swimmers. It just depends on the swimmer. From personal experience, bromamines are less corrosive to natatoriums than chloramines and are less noxious. But they still exist and should be appropriately ventilated out of indoor pools.
Trihalomethanes (THMs)
The bigger issue in bromine pools is actually Trihalomethanes (THMs). Only one of the four trihalomethanes (chloroform) can be created in chlorine pools because bromine is not in the water. But if you are using chlorine to recharge bromine or using a product like BCDMH (which contains both bromine and chlorine), all four types of THMs are possible in your water:
Trihalomethanes are formed when chlorine and/or bromine oxidize hydrocarbons in the water. They are disinfection byproducts (DBPs) and contribute to bad air quality and swimmer irritation. It's best to avoid them if at all possible. According to Richard Falk and the EPA, THMs are likely carcinogenic, especially the brominated THMs. Chloroform, however, would take much higher exposure levels.7
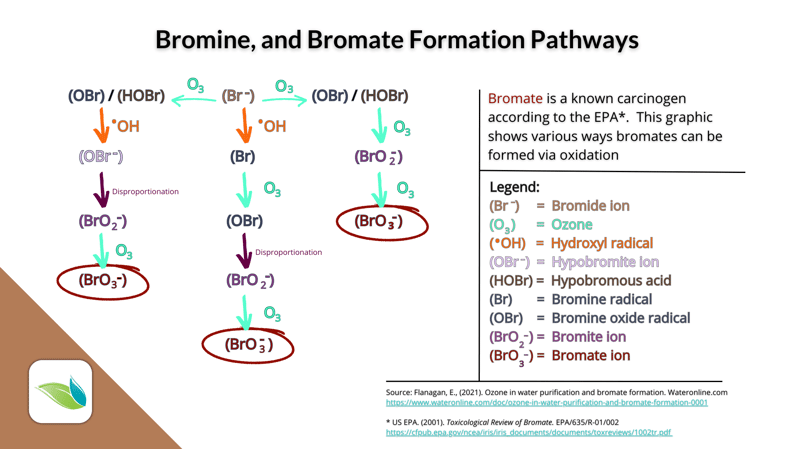
Bromates

Perhaps the most important byproduct of bromine is created if bromine is exposed to direct sunlight or UV systems. It's called Bromate (BrO3-). Bromate ion is classified as a known carcinogen by the EPA.
Here's an excerpt from the EPA's decision on Halohydantoins:
"EPA has determined that there are potential risks of concern for the outdoor swimming pool use pattern of halohydantoin products that contain bromine. The risks only exist for outdoor swimming pools because the bromate ion is formed under UV light exposure." - pg. 9 8
The EPA's report goes on to say:
"In this interim decision, the agency is requiring that all halohydantoin products that contain active ingredients with bromine (PC codes 006315, 006317, 006322, 006333 and 128989) remove the outdoor swimming pool use and add label language for outdoor hot tubs and spas." - pg. 12 8
Studies like this are why we recommend against using bromine in outdoor pools and spas (unless the spa is covered when not used). And it's not ONLY outdoor pools, like page 9 in the above quote suggests. Bromates are also formed in any pools or spas that have UV systems, ozone (O3), hydrogen peroxide (H2O2), or Hydroxyl radicals (•OH). With ozone, for example, the bromide ion (Br-) gets recharged only 77% of the time, while the other 23% of the time, it gets oxidized into the harmful Bromate ion (BrO3-).9
What secondary disinfection systems can be used on a bromine pool?
We at Orenda support secondary disinfection systems on chlorine pools. They serve an important function, especially for high-use commercial pools. Unfortunately, most of these secondary disinfection systems are incompatible with bromine pools because they create bromates.
- Ozone (O3) and hydroxyl radicals (•OH) can create bromates via oxidation.10
- UV systems and direct sunlight create bromates via photolysis.11
Drinking water and wastewater facilities are using bromine less and less because of the limited options for oxidation without creating bromates.12
That leaves only a few options for secondary systems, none of which are registered as disinfection systems. The first type of secondary system is Hyperdissolved Oxygen (HDO). The second is Hydrodynamic Cavitation (HDC). We have not conducted studies on how well these systems perform in bromine pools, but as far as we can tell, they are compatible with bromine.
Is Bromine compatible with Chlorine?
Generally speaking, bromine is not really compatible with chlorine...but this deserves clarification.
Yes, if you use chlorine products in a bromine pool. If you have a bromine pool or spa and you are looking for a way to recharge bromide ions, a chlorine shock can be used. And yes, if your bromine product of choice is BCDMH, it contains chlorine. Chlorine can recharge bromide ions into HOBr and keep the bromine cycle going, and in that context, chlorine is fine to use in a bromine spa or pool. In fact, it's arguably the product of choice for recharging bromine because chlorine will not produce bromates.
No, if you use bromine products in a chlorine pool. If you have a chlorine pool or spa, we do not recommend using bromine products. We'll use the most common example of sodium bromide algaecide. Sodium bromide is a salt (NaBr) that dissociates into sodium and bromide ions. Then the chlorine in the pool recharges the bromide ion into HOBr, the actual disinfectant. But the key is the bromide ions are left in the water. These bromides will build a permanent chlorine demand cycle.
Bromine can take over a chlorine pool
Because bromine regenerates, it stays in the water long-term in various forms. The continual use of bromine products will eventually convert a chlorine pool into a bromine pool. This conversion happens when the water has more bromide ions (Br-) than free chlorine. It only takes a few ppm of bromide to turn a chlorine pool into a bromine pool.11 The only practical way to revert the pool to a chlorine pool is to remove the bromides from the water, which usually means a complete drain and refill. Bromides can also be removed via reverse osmosis filtration and activated carbon filters.
Bromine can also be reduced through bromamine off-gassing, though this process is very slow and not recommended.
So how do you know if your chlorine pool has become a bromine pool? In an outdoor pool, the first symptom is a severely-increased chlorine demand. This is due to chlorine's affinity for oxidizing bromide ions (Br-), which are easier for chlorine to oxidize than other contaminants. Temporarily that's fine, except it creates HOBr, which cannot be protected from sunlight degradation, so it's gone quickly. This process dominates chlorine demand.
You can also distinguish between bromine and chlorine using a DPD test in a bucket of pool water with household ammonia, as outlined in this PCTI tech bulletin.13
Conclusion
Both chlorine and bromine are halogen elements that can disinfect water and oxidize non-living contaminants. Chlorine is comparatively stronger, as it is one row above bromine on the Periodic Table of Elements. This also means chlorine (and other oxidizers) can oxidize bromide ions and convert them into hypobromous acid, which "recharges" bromine. This is one nice feature of bromine. But it's a two-edged sword.
If bromide ions (Br-) get oxidized enough (by ozone, AOP, or hydrogen peroxide), they can also form harmful bromates. Bromates can also be created by UV light (either a UV system or direct sunlight). And since bromine does not have a stabilizer against UV degradation (unlike chlorine, which has cyanuric acid), bromine should not be used in outdoor pools or indoor pools with UV systems.
Bromine has been sold as a decent option if you have a spa with a low-to-moderate bather load. It performs well in spas, and its bromamine byproducts are less irritating and corrosive than chloramines. But there is an abundance of scientific research about the consequences of bromine, and we believe bromine products are likely to be outlawed in the future. Until then, if you have an indoor bromine pool, we recommend oxidizing with potassium monopersulfate or liquid chlorine. These oxidizers can recharge bromine without creating bromates.
We hope you found this article informative and helpful.
1 Lowry, Robert W. (2018). Pool Chemistry for Residential Pools. Ch. 14 (pg. 169). Pool Chemistry Training Institute.
2 Lowry, Robert W. (2018). Pool Chemistry for Residential Pools. Ch. 14 (pg. 172). Pool Chemistry Training Institute.
3 In the halogen group of elements on the Periodic Table, higher elements can oxidize lower elements. Chlorine can oxidize bromide ions into bromine, and both bromine and chlorine can oxidize iodide ions into iodine. Theoretically, fluorine could oxidize chloride ions into chlorine, but according to this source, fluorine is too powerful of an oxidizer that it actually oxidizes water itself, so aqueous reactions cannot take place. This is why fluorine is not used in swimming pools, and the next-best halogen (chlorine) is the most dominant residual sanitizer on the market.
4 Lampre, I., Marignier, J.L., Mirdamadi-Esfahani, M., et.al. (2013). Oxidation of Bromide Ions by Hydroxyl Radicals: Spectral Characterization of the Intermediate BrOH•-. Journal of Physical Chemistry. 117, 5. (pp. 877-887). https://doi.org/10.1021/jp310759u
5 Mensah, A., Berne, F., Allard, S., et.al. (2022). Kinetic modelling of the bromine-ammonia system: Formation and decomposition of bromamines. Water Research, vol. 224. https://doi.org/10.1016/j.watres.2022.119058
6 Ozone can "recharge" bromine by oxidizing bromide ions into HOBr. But ozone and AOP can also convert bromide ions into harmful bromates. This is complex chemistry, but in summary, ozone can oxidize bromide ion (Br-) into hypobromous acid (HOBr) and the hypobromite ion (OBr-). But if more ozone continues to oxidize HOBr or OBr-, bromite ion (BrO2-) and eventually bromate ion (BrO3-) will be created. A good explanation and graphic of this process can be found here.
7 "Of the four trihalomethanes, only the brominated ones are effectively carcinogens since chloroform is only carcinogenic at cytotoxic levels that are rather high." - Private email with Richard Falk, June 2023. EPA fact sheet: bromodichloromethane.
8 US EPA. (2021). Halohydantoins: Interim Registration Review Decision, Case Number 3055. (pp. 9-12) US EPA docket# EPA-HQ-OPP-2013-0220.
9 Rice, Rip G. (1995). Chemistries of Ozone for Municipal Pool and Spa Water Treatment: Facts and Fallacies. Journal of the Swimming Pool and Spa Industry, vol 1. (1). (pp. 25-44). -- This specific reference is on page 36.
10 Haag, W. R., & Hoigne, J. (1983). Ozonation of bromide-containing waters: kinetics of formation of hypobromous acid and bromate. Environmental Science & Technology, 17(5), 261-267.
11 Fang, J., Zhao, Q., Fan, C., et.al. (2017). Bromate formation from the oxidation of bromide in the UV/chlorine process with low pressure and medium pressure UV lamps. Chemosphere, 183, (pp. 582-588).
12 Kishimoto, N., Nakamura, E. (2012) Bromate Formation Characteristics of UV Irradiation, Hydrogen Peroxide Addition, Ozonation, and Their Combination Processes. International Journal of Photoenergy, vol. 2012.
13 Lowry, Robert W. (2019). Bromine or Chlorine: What's in Your Pool? Pool Chemistry Training Institute, Tech Bulletin.

-1.jpg?width=800&height=450&name=trihalomethanes%20(THM)-1.jpg)