What is a Hot Start?

A "hot start" is a method for starting up a new or resurfaced pool using a lot of acid. It is extremely aggressive–by design–so it etches calcium off the surface, hopefully evenly. The objective of a hot start is to "burn up" plaster dust and any imperfections left behind by the plaster application process, so that the customer is left with a beautiful looking pool. A hot start may also be called a "zero alkalinity startup" or an "acid startup". This article will discuss what hot starts are, why they are used, how they are done, and the chemistry behind the process.
Hot Starts are primarily used for cosmetic exposure of aggregate
There are many ways to start up a brand new (or resurfaced) pool. We promote our Orenda Startup™, which we believe is the least intrusive and most effective way to start up a pool. Adding chelated calcium early and balancing the LSI protects the integrity of the brand new pool plaster, quartz or pebble finish, and can prevent plaster dust. That being said, one thing the Orenda Startup™ does NOT do, is expose aggregate (like beautiful pebble or quartz). So let's talk about what plaster exposure means, and the ways to do it.
Exposure of pool finishes
 To let the beautiful aggregate shine and look great in the pool, it is very often necessary to 'burn off' some of the cement that binds the surface together. The cement is calcium based. During the application process, a thin film of calcium can rise to the surface and make the entire surface look hazy, gray or white. On gray or darker surfaces, trowel marks and other lines can be visible.
To let the beautiful aggregate shine and look great in the pool, it is very often necessary to 'burn off' some of the cement that binds the surface together. The cement is calcium based. During the application process, a thin film of calcium can rise to the surface and make the entire surface look hazy, gray or white. On gray or darker surfaces, trowel marks and other lines can be visible.
The photo here shows trowel marks and discoloration because of it. We asked some plaster experts around the country to look at the photo, and they all said it looked like "water troweling". When the pool looks like this, an exposure is necessary to even out the look of the finish.
There can also be puddle marks and other unsightly discolorations on a new surface. This film is called many different things: cream, butter, chalking, and haze (and there are probably many more nicknames for it, but let's just refer to this film as cream). The cream needs to be removed, or else the surface will look bad, and nobody wants that. There are primarily three ways to do it.
1. Acid Washing
For quartz and pebble finishes especially, acid washing is a key part of the pool finishing process. The basic idea is to take very diluted muriatic acid and wash the entire surface with it, quickly. Then neutralize with soda ash (not sodium bicarb!). The acid will burn off the top layer of cream, exposing aggregate below for what is hopefully a beautiful and evenly colored finish. But there are many risks and ways that acid washes can go wrong. Actually now that we mention that, we should probably write an article just about startup failures and how they can be avoided.
 A bad acid wash can cause streaking lines, discolorations and ugly, irregular shaped marks, and generally uneven looking finishes. For example, if the acid is poured too high and touches tile grout, the grout that is burned can streak down in thin lines down the surface, leaving obvious marks. On tanning ledges (also called sun shelfs), it is common to see rings and marks that look like white puddles. How about a white bowl in the bottom of the pool, or rings around the main drains? These are signs of acid leaving marks. Either they were not rinsed and neutralized properly, or the acid was too concentrated, or it was on there for too long. Who knows.
A bad acid wash can cause streaking lines, discolorations and ugly, irregular shaped marks, and generally uneven looking finishes. For example, if the acid is poured too high and touches tile grout, the grout that is burned can streak down in thin lines down the surface, leaving obvious marks. On tanning ledges (also called sun shelfs), it is common to see rings and marks that look like white puddles. How about a white bowl in the bottom of the pool, or rings around the main drains? These are signs of acid leaving marks. Either they were not rinsed and neutralized properly, or the acid was too concentrated, or it was on there for too long. Who knows.
With a bad exposure, it is very difficult to clean up the mess. And no, the Orenda Startup will not clean up a bad exposure.
Neutralizing during and after an acid wash is critically important
One of the biggest mistakes we see when it comes to acid washing is a failure to neutralize enough, or fast enough. Acid will continue to dissolve calcium, so it needs to be neutralized after it has done its job. The sooner, the better. Every plaster company has their own methodology to this it seems, but all of them know they need to neutralize the acidic water that collects in the bottom bowl of the pool. But there's a problem: many acid wash crews neutralize with baking soda (sodium bicarbonate). Sodium bicarb has a pH of about 8.3-8.6, which is usually not basic enough to neutralize diluted muriatic acid used in an acid wash.
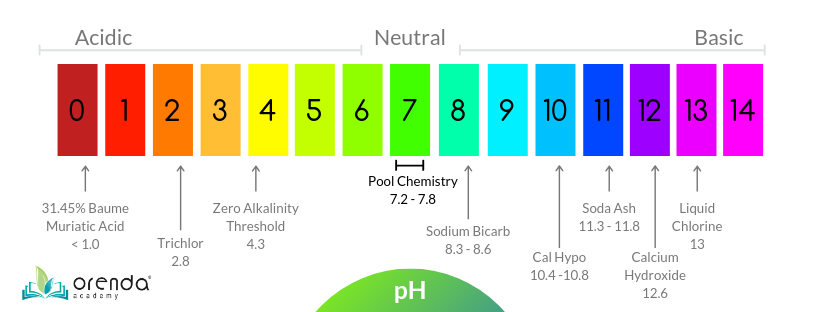
As you can see from the chart, the type of muriatic acid we use in the pool business, 31.45% Baume, is less than 1 on the pH scale! It has to be heavily diluted for acid washing. But look over on the other side at Sodium bicarb, which has a pH between 8.3 and 8.6. That is not nearly alkaline (or basic) enough to counteract the strong muriatic acid, even when diluted. Since the pH scale is logarithmic, 11.3-11.8 pH soda ash is roughly 1000x more basic than sodium bicarb. Using bicarb may not be enough, and when acidic water sits in the pool long enough, it can leave behind a white bowl, where calcium is pulled to the surface and carbonates. It can be ugly.

So use soda ash when neutralizing an acid wash, as seen in this photo:

When done right (and neutralized right), acid washing can leave behind a beautiful pool finish.
2. Polishing
Certain finishes are designed to be mechanically polished until smooth. The material is applied and troweled just like any other finish, but instead of acid washing, the crew pulls out electric polishing tools and sands down the finish so it is very smooth. This method is more labor intensive and time consuming, but also puts more control in the hands of the applicators, as opposed to an acid wash. Polishing physically grinds down the cement and aggregate.

In some cases, an acid wash is used even after polishing, but from what we are told, it is an even lighter concentration than a normal acid wash. It depends on the manufacturer and the applicators preferences. Polishing is not applicable to all finishes, like pebble finishes that are meant to have round pebbles exposed. Polishing would grind them all flat and smooth.
As with any exposure technique, polishing has its risks too. It's difficult to get a grinder into nooks and corners, so the polishing will have spots that are rougher than others. Sometimes polishing can leave behind visible marks and circular lines. Nothing is perfect, but by and large, polished finishes tend to look great.
3. Hot Start (or Zero Alkalinity Startup)

Ah yes, the focus of our article: the hot start. A hot start is when you fill up the pool with water, then put A LOT of acid in to lower the pH significantly. From what we are learning, the best practice for a hot start is to suspend circulation pumps into the middle of the pool (for water movement), but many people just pour acid straight into the pool.
We have heard some crazy numbers, and what's funny is everyone we talk to seems to have a different opinion. Some people say "1 gallon of acid for every 5,000 gallons of water." Others say "1 gallon for every 2,000 gallons of water". Whatever it is, it's usually more than 4 gallons of straight muriatic acid for a typical backyard pool. Any way you look at it, it's a lot of acid.
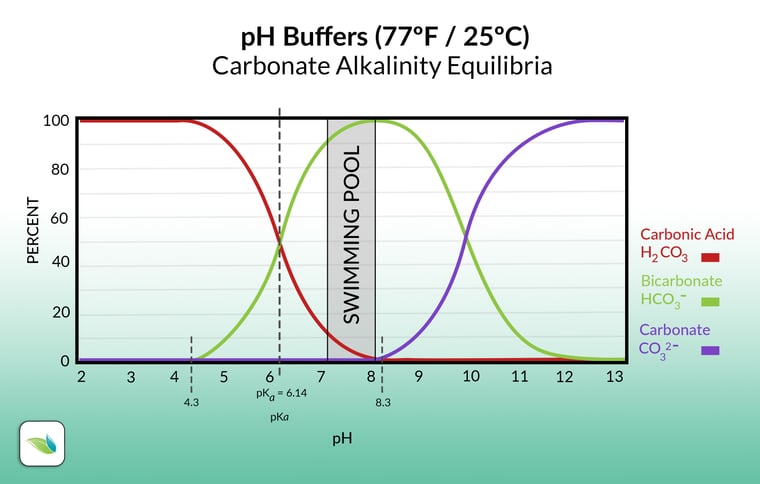
The objective of a hot start is to get the pH of the pool down to 4.3 or below so it burns up the plaster dust and [supposedly] exposes the surface evenly. Why 4.3 pH? Because 4.3 pH and below means there is zero alkalinity in the water. Hence the term "zero alkalinity startup". Without any alkalinity, water can dissolve a lot of calcium from the surface quickly, and for a prolonged period of time. Here's the alkalinity equilibrium chart, showing that 4.3 pH and below has no alkalinity, just carbonic acid:
By getting the water below 4.3, it is super aggressive, intentionally. The acid etches the outermost surface of the pool to deliver an evenly exposed finish (hopefully). After a few days, the water is neutralized with soda ash (again, not sodium bicarb!) and the rest of the traditional startup process can proceed. Calcium hardness will drift up because the acid dissolves so much out of the surface and pulls it into solution.
Some experts we have talked to say the super low pH "oxidizes the pigment" which can make a pool surface darker. While that's not chemically accurate, we get the basic idea of what they mean. Hot starts can definitely make a pool appear darker than it was.
Problems with hot starts
There are many issues with hot starts, but let's start with some facts about how plaster cures. Calcium hydroxide (Ca(OH)2) is the most soluble form of calcium, and takes longer to cure than other components in plaster. According to the National Plasterers Council, this can take at least 28 days, and more like 30-60 days. It can take YEARS for all of the cement to cure completely, but the vast majority is done within the first 28 days. Calcium hydroxide, because it is so soluble, is a very important component in the cement. It fills in cracks and voids around aggregates, then carbonates into hardened calcium carbonate (CaCO3). That is to say, calcium hydroxide is a good thing to have, because when it hardens, it reinforces the cement that holds the plaster together.
But what if that calcium hydroxide is stolen?
Unfortunately, aggressive water steals calcium hydroxide, because water always seeks its own equilibrium on the LSI. When you do a hot start, it's not just aggressive water...it's SUPER aggressive water with zero alkalinity. The amount of calcium hydroxide that is stolen can be astounding. Sure, you won't see much plaster dust (because the acid burns it up so fast), but rest assured, you are losing a lot of calcium from your surface when you do a hot start. Hot starts prioritize cosmetics over the integrity of the surface. We at Orenda prefer to protect the plaster as much as possible.
What is the preferred method of exposure?
Each method has its benefits and drawbacks. Acid washing allows more control of the exposure, but risks visible lines, rings, and an ugly look. Polishing is great, but is difficult to get into every nook and corner, and sometimes grinding marks are left behind. Hot starts may give the most even exposure, but they etch the surface making it rougher, and often times, darker. Furthermore, hot starts take a lot of valuable calcium from the cement, which cannot be put back.
At Orenda, we strongly advise against hot starts as a practice, especially in the first 30 days. This is based on field experience of countless plaster experts we have spoken with, as well as the technical information available from the National Plasterers Council. We also consistently look for contrarian opinions and interpretations of research, which have helped us form our opinions too. We don't like hot starts because they are just too aggressive. Hot starts pull out valuable calcium hydroxide from the finish, which can compromise the longevity of the surface itself.
If an acid wash is necessary, try to make it as light as possible, for as little time as possible, and neutralized as thoroughly as possible. For polished surfaces, hire an experienced polish crew that takes their time to do it right. Nothing will be perfect, but get as close as possible.
Remedies for bad exposures
We see it all the time, and people send us photos and videos of bad looking pools. Problems can happen, but fortunately, most of them are just cosmetic. For example, if the acid wash was done poorly or not neutralized properly, there can be white marks in the bottom of the pool, or streaking lines. Sometimes the best answer is to simply drain the pool and do it again, but do it right (i.e. repolish or do another light acid wash, but better). If draining is not an option, experts tell us the most viable solution is an acid treatment sometimes called an "acid bath" or a "no drain acid wash".
 White deposits like puddle marks and streaks do not always mean the surface is damaged. Often times you're just looking at a concentration of carbonated calcium trying to run its course during the curing process. These marks are usually just cosmetic blemishes, not material failures or flaws.
White deposits like puddle marks and streaks do not always mean the surface is damaged. Often times you're just looking at a concentration of carbonated calcium trying to run its course during the curing process. These marks are usually just cosmetic blemishes, not material failures or flaws.
This acid bath remedy is different from a hot start, because you are not dropping the pH low enough to wipe out all alkalinity (< 4.3 pH). Instead, the pH goes to about 6.5 - 6.8, which is slightly below neutral 7.0, but enough to restore color and even out the cosmetic look of a pool finish. It can take days and a lot of brushing, but with the help of SC-1000, the calcium that is dissolved from the surface will go into solution and stay there. It's not ideal, but it's far less aggressive than a hot start.
Hopefully you do not encounter the need to do any of these remedies, as they are laborious and not fun. That said, they can restore color and the look of a pool, provided they are done correctly. We at Orenda care first and foremost about the quality of the surface and its integrity, so when remedies are needed, be ever mindful of the homeowner's property. Do not over-do it and cut corners. Take the time to do it right.