Understanding Borates in Pool Chemistry

The use of borates in swimming pools dates back several decades, though the recent popularity of borates makes them seem new and cutting-edge. While we at Orenda do not endorse the use of borates, they are becoming more widely used. Because borates impact the LSI, we have included borates in our Orenda App 3.0 update. And since borates are in our calculator, we're here to explain what they are and how they work in this article.
This article does not contain dosing information, and neither does the Orenda App. If you choose to use borates, consult dosing information elsewhere.1
Covered in this article:
- What are borates?
- Limits and staying power
- What do borates do in swimming pool chemistry?
- pH buffering
- pKa values
- The impact of Borates on the LSI
- Borates and the pH ceiling
- Algaestat claims
- Lower chlorine demand
- Improved water clarity
- pH buffering
- Types of borate products
-
- Labels and certification
- Borate products
-
- Impacts on health and safety
-
- Are borates illegal in California?
-
- Conclusion
What are borates?
According to our primary sources2, borates and boric acid are forms of boron that are used in many products like household cleaners, laundry detergents, and insecticides. In swimming pools, borate products are primarily used as a pH buffering system in water; specifically to buffer against the increase in pH. More on that in a moment.
Because of their pH buffering capability, borates impact the Langelier Saturation Index (LSI), particularly when the pH is high. Borates' LSI impact is pretty small compared to Cyanuric Acid (CYA), but it's enough that we have included it as an optional factor in the Orenda Calculator.
Limits and staying power
Every reputable source we read and spoke with agreed the maximum borate level in a pool is 50 ppm. They are most effective between 30-50 ppm, much like cyanuric acid. And the more borate you have in your water, the less exposure (or consumption of pool water) is needed for adverse health effects. More on health and safety later in this article.
Borates also stay in the water for a very long time. Borates, like CYA, do not evaporate out of the pool, so they tend to stay at approximately the same level most of the year. Once you have your borate level established, you only lose it from backwash, splashout, or other water loss. And since no other pool chemicals that we know of contain borate (unlike stabilized chlorines containing CYA), accumulation should not be an issue.
What do borates do in swimming pool chemistry?
If you do your own research online (like we did), you will see dozens of opinions about borates. And while we selected the five most reputable primary sources we could find, we also read secondary sources to hear everyone's opinions. Basically, the benefits of borates can be boiled down to these four things:
- Buffer pH from increasing
- Limit algae growth (algaestat)
- Lower chlorine demand
- Improve water clarity
Let's dive deeper.
pH buffering
The carbonate alkalinity system is the dominant buffering system used in swimming pools, and it buffers against changes in pH, especially against reducing pH. Borates are an entirely different buffering mechanism. And when we say “buffering”, we mean the chemistry resists change in pH.
Here's an excerpt from the journal article "Boron Separation Processes":
"Boric acid is very weak and does not dissociate in aqueous solution as a Bronsted acid but it acts as a Lewis acid by accepting a hydroxyl ion to form the tetrahydroxyborate ion..." 3
B(OH)3 + H2O ⇄ B(OH)4- + H+
Boric acid + water ⇄ Tetrahydroxyborate + Hydrogen4
Bicarbonate is the conjugate base of carbonic acid. It buffers against reducing pH by taking or giving away a Hydrogen ion (H+). Carbonic acid is a "Bronstead acid" that dissociates in water and loses a Hydrogen ion.
Borate is the conjugate base of boric acid. Boric acid buffers against increasing pH by taking or giving away a Hydroxide (OH-). Boric acid is a "Lewis acid" that does not dissociate in water; instead, it associates with a Hydroxyl ion.
Both of these buffering equilibriums have an optimal pH where they can resist change in pH. On the graph below, you can see the optimal buffering pH is where the lines intersect. This is where 50% of the acid is present along with 50% of its conjugate base. This point is called the “pKa” value.
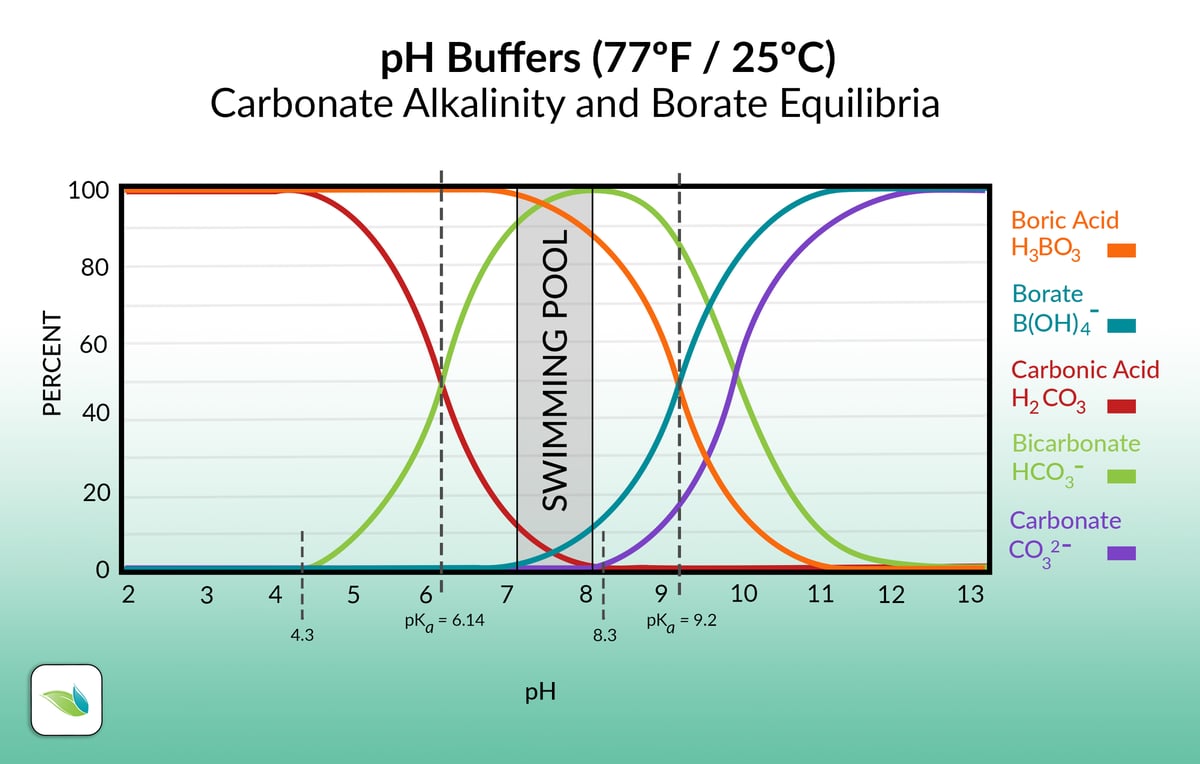
pKa values
As mentioned above, the pKa value is where the buffering system can resist change in pH the best. It's where the acid and its conjugate base intersect. Look where boric acid and borate intersect on the graph above. And this is important because you can think of these points as a kind of boundary for pH to be contained within. The closer your pH gets to the pKa value, the more resistance it faces.
The pKa value depends on water temperature, but it doesn’t change all that much. Multiple reputable sources online give slightly different pKa values for bicarbonate and borate. So to simplify, we will use the numbers at a water temperature of 77ºF (25ºC). That seems to be a standard temperature that the sources liked to use. At 77ºF/25ºC:
Now let's apply this to swimming pools. As your pH naturally rises, the closer it gets to 9.2, the more resistance borates will provide. This helps to slow the rise in pH, making it easier to contain pH.
In our opinion, if you already are using Henry’s Law to your advantage you can contain pH without borates because you can already determine your pH ceiling. That being said, borates can make the job easier and give you an extra layer of buffering.
Borates' impact on the LSI
Because boric acid/borates buffer pH, they contribute in a small way to total alkalinity. The higher the pH, the more important it is to adjust for them. Remember that the LSI calls for the carbonate alkalinity, which is just the carbonate/bicarbonate buffering system. To calculate carbonate alkalinity, we must deduct cyanurate alkalinity and borates.
The Orenda Calculator does the math for you, just like it takes CYA into account. If you do not see Borate on your calculator screen, go to:
Orenda app > main menu > app settings > select borates > done.
Then borates will show up on your calculator screen as a 7th LSI factor. Provided you are not overdosing borate (always stay 50 ppm or less), the LSI impact is small, but at higher pH it matters more.
Borates and the pH ceiling
Because borates contribute to total alkalinity, just like CYA, the carbonate alkalinity is lower (relative to total alkalinity). This means the pH ceiling is reduced when you have borates in your pool.
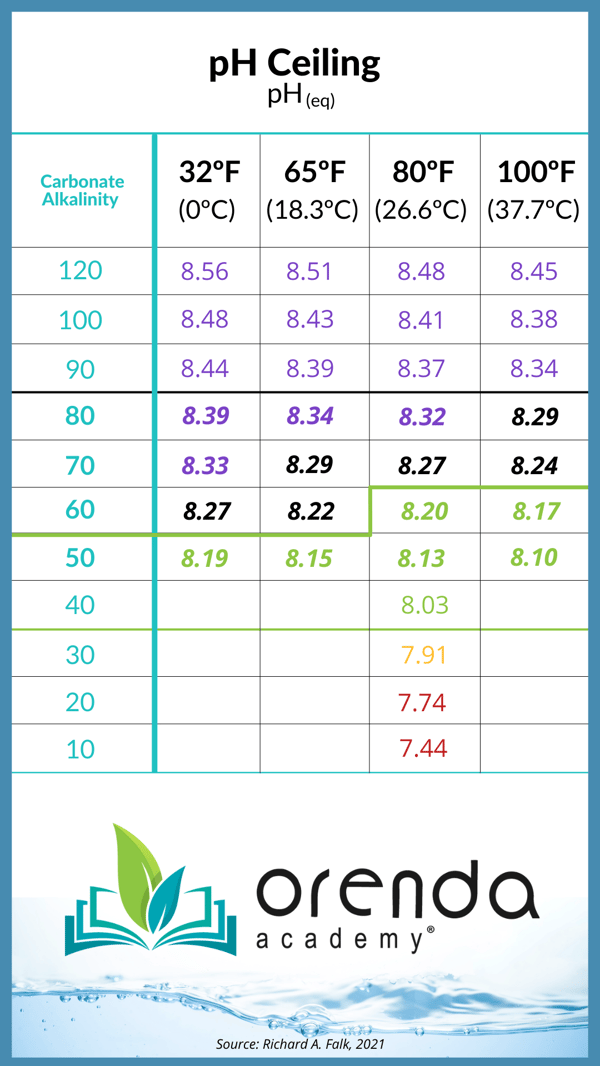
The Orenda App (version 3.0 and later) will show you this in real-time. When you select "show" secondary factors, you will see the carbonate alkalinity and pH ceiling on the calculator. You will notice when you increase borate on the Orenda Calculator, the LSI, carbonate alkalinity, and pH ceiling all decrease. Play around with the app to see the impact that borates have on your chemistry.
Algaestat claims
At least a dozen articles and forum posts online talked about how borates are algaecides and algaestats. Opinions on the matter range from "borates remove CO2 from the water and starve algae" and "borates are herbicides" to "borates suppress pH to keep it lower so chlorine is more effective at killing algae."
Yeah, there's a lot to unpack here. And we took the time to read every word of every post we saw about it. We'll save you some time: none of those theories are true.
One of the most reputable sources on the topic is the late Robert Lowry, arguably the most influential educator the swimming pool industry has ever known. According to Lowry's textbook Pool Chemistry for Residential Pools:
"So how do borates prevent algae? Borates disrupt cell wall development, metabolism and cell division. So borates are good perhaps at preventing algae (algaestatic) rather than killing algae (algicidal). You will need less chlorine (have a lower chlorine demand) when using borates because you are preventing algae. 50 ppm borate lowers the free chlorine recommendation from 7.5% to 5%." - Robert W. Lowry, pg. 59. 7
So borates are not an algaecide, but according to Lowry, they can help prevent algae reproduction (at least one Sodium Tetraborate product does have an EPA registration as an algaestat). Slowing the growth/reproduction rate, as we cover in-depth in Pillar 3, can improve chlorine efficiency because sanitization is a race between the kill rate of the sanitizer and the reproduction rate of the contaminant.

Lower chlorine demand
This claim ties into the previous two claims about pH buffering and algae prevention. Some sources online think that the suppressed pH means stronger chlorine (which would not apply in pools with CYA). Others say it's because of borates' algaestat properties. If that's true, it's a logical idea. But experts disagree on how much chlorine savings borates can actually provide.
The claim is up to 30-50% chlorine savings, but that's theoretical at best. In the real world, as pointed out by Kim Skinner from onBalance:
"Adding borate will NOT reduce chlorine costs by 30% to 50% as is being claimed. It is generally known that residential pools lose, on average, about 1 to 2 ppm of chlorine per day. According to many service companies, residential pools generally lose the same amount of chlorine daily whether they contain borate or not. And the suggestion to maintain lower chlorine levels is risky and problematic." 8
We agree with Skinner that maintaining lower levels of chlorine could be risky and problematic. Water needs a minimum sanitizer level to stay sanitized.
We also spoke with Richard Falk about chlorine savings, and his answer was also clear:
"There are NO chlorine savings at all from using borates or boric acid IF you maintain a high enough active chlorine level (FC/CYA ratio) such that chlorine is able to kill algae faster than it can reproduce. The ONLY chlorine savings is if you aren’t using a high enough active chlorine level. In that case, algae can grow about as fast or faster than chlorine can kill it, and in that situation there will appear to be chlorine demand as it has more algae to kill. - Richard Falk 9
To paraphrase Richard, if you already have enough chlorine to stay ahead of the reproduction rate of algae, you will not notice chlorine savings, because it's being used up properly. The savings would occur if your chlorine falls behind (which is most pools, let's be honest). But even so, the chlorine savings would be minimal.
But outside of that, we do not have enough information to form a worthwhile opinion. We have not heard customers of ours talking about major chlorine savings on their borate pools. And in the pool industry, claims of chlorine savings are usually overstated.
Improved water clarity
This one is subjective. Water clarity has to do with filtration, circulation, and chemistry. Richard Falk also thinks it could have something to do with reduced surface tension. Clarity and "sparkle" are about how light penetrates and reflects in the water. If more light can get into the water, or if it reflects better, the water can appear cleaner and clearer. We know this because enzymes also reduce the surface tension of water and improve water clarity.
Honestly, we don't know if borates provide improved water clarity directly or indirectly. None of the sources online cited any studies about this. It was all anecdotal. And anecdotal evidence is often compelling, it's just not measurable.
Types of borate products
Labels and Certification
Before going further, Orenda strongly encourages only using products that are specifically labeled for use in swimming pools in accordance with federal, state, and local laws. Part of that criteria is dosing information. The CDC's Model Aquatic Health Code (MAHC) calls for any additive used in commercial swimming pools to be either EPA Registered, NSF-50 (recreational water), and/or NSF-60 (drinking water) certified.10
It stands to reason that this same safety standard should be applied in residential pools too. If you are using any product that is not specifically labeled for use in swimming pools and/or lacks those certifications, we advise against using those products in your pool.11
Borate products
There are several borate products you can get on the market, though they may not be available in pool stores (especially in California). The three most common products are borax, which is sodium tetraborate decahydrate (Na2B4O7.10H2O), sodium tetraborate pentahydrate (Na2B4O7.5H2O), and boric acid (B(OH)3).
There are a couple of other variations, but from what I've read, they're not nearly as popular. Apparently, the big difference between tetraborate products and boric acid is the pH. Tetraborate products have a high pH and require acid to neutralize them when added into the pool, whereas boric acid needs no pH correction when it's added to water. Learn more about dosing borate chemicals here.
Borate products are dry, granular chemicals.
Impacts on health and safety
Many of the questions we hear about borates are about their toxicity and health impacts on water. It's true that Borates have been banned in California in the past, and there have been several health studies–including one published by the EPA–on boron's health impacts on drinking water.12
According to Richard Falk, toxicity is mostly a concern for dogs drinking pool water, but they would need to drink a lot of pool water to get sick, assuming the pool is at 30-50 ppm borate. Humans would need to drink even more, depending on body mass. From what we are reading, the main reason for the 50 ppm maximum recommendation is health and safety, to prevent long-term exposure problems such as reproductive risks in young males. 50 ppm gives benefits of borate with low risk of exposure issues.13
Are borates illegal in California?
Let's cite back to Robert Lowry again. His published information on borates is perhaps the most concise, yet detailed source out there. According to Lowry, borates are not illegal in California (you can buy Borax at the grocery store, and boric acid at a garden store), but they cannot be sold in pool stores. That's because of how borate products are classified by the California EPA:
"The State of California has a unique law with regard to what they define as "water modifiers" under a pesticide category of "spray adjuvants." California EPA says that if a product is used to modify the pH of water to maximize the performance of a pesticide (proper pH and alkalinity improves the performance of chlorine) then it is subject to all the same regulations as a pesticide. So baking soda and borates are required to be registered as a pesticide in California and all toxicity, cancer and exposure tests must be done prior to approval and then the companies selling these products have to pay a use fee to the State for every pound sold. Baking soda has complied, but not borate." 14
In other words, bureaucracy at its finest. You can buy the products at other stores, but not pool stores.
Conclusion
Borates are growing in popularity for pool care. The main benefit of borates is that they help buffer the pH from rising. According to Robert Lowry, they also slow the growth of algae, which can improve chlorine efficiency. Other benefits depend on whom you're asking. Borate users have their own opinions on how they have impacted their pools. Practical first-hand knowledge is probably more valuable than purely theoretical numbers.
The purpose of this article was to explain what borates are and what they do in pool water. We neither encourage nor discourage the use of Borates. We are only including them in the Orenda app because of their impact on the LSI. We did not include dosing information in our app, nor in this article. If you choose to use borates, use 50 ppm or less, which is the recommended amount to minimize exposure risks.
In our opinion, borates may be beneficial, but if you contain pH with lower carbonate alkalinity (which reduces your pH ceiling to a manageable level), we do not think they are necessary. But again, we encourage you to form your own opinion and make an informed decision. We hope this article helps you do just that.
1 Here are two resources with borate dosing information: PHTA and Pool Chemistry Training Institute.
2 Researching this article involved over 30 websites, peer-reviewed journals, and other chemistry publications. Upon further reading, of those 30+ open tabs on our browser, most of them were just paraphrasing (or outright plagiarizing) only a handful of original sources. Those main sources are the basis for this article. They are:
- Robert W. Lowry, Pool Chemistry Training Institute
- Kim Skinner and Que Hales, onBalance
- Richard A. Falk, whom we cite in virtually every chemistry article
- John Wojtowicz, 2001. Swimming Pool Water Buffer Chemistry. JSPSI Vol. 3 #2, pp. 34-41.
- A slew of articles on ScienceDirect.com
3 Kochkodan, Hilal, et.al. (2015). Boron Separation Processes. §2.3.2 Dissociation of Boric Acid in Water.
4 This confused us because there are reputable sources that have Boric acid written in two different ways. Some say Boric Acid is expressed as B(OH)3, while others have it as H3BO3. Technically they're the same. But according to this thread, the reason it's expressed as B(OH)3 is because unlike most acids, which dissociate in water, boric acid is associated with water.
5 University of Arizona Biology Department (2004). Bicarbonate as a Biological Buffer.
6 Kabay, Bryjak, et.al. (2015). Boron Separation Processes. §9.2. Boron Chemistry.
7 Lowry, Robert W. (2018). Pool Chemistry for Residential Pools. Ch. 7, pg. 59.
8 Skinner, Kim (2020). Interview for the article "Borates stabilize pH, reduce algae growth". Service Industry News.
9 Copied/pasted from an email conversation with Richard Falk during his review of this article pre-publication.
10 NSF.org (2019). Navigating the Chemical Requirements of the Model Aquatic Health Code.
11 CDC.gov (2019). Using the MAHC. Model Aquatic Health Code (§ 5.7.3 Disinfection and Water Quality).
12 US Environmental Protection Agency. (2008). Drinking water health advisory for Boron. Pages 13-24.
13 Falk, Richard A. (2014). Are Borates Safe to Use? Posted on the TFP forum.
14 Lowry, Robert W. (2018). Pool Chemistry for Residential Pools. Ch. 7, pg. 68.
